 
The aluminum anode reacts with oxygen from the air to. 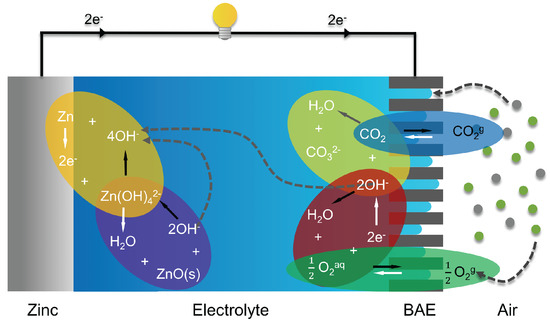
It is hoped that the Review will stimulate scientists and engineers to develop more reliable aluminum batteries.Ĭoulombic interaction electrolytes interface reactions rechargeable aluminum batteries. The heat released from an aluminum-air battery has a great effect on its performance and operating life during the discharge process. The Aluminum-Air Battery is a type of battery that uses an aluminum anode and an oxygen cathode. To provide a good understanding of the opportunities and challenges of the newly emerging aluminum batteries, this Review discusses the reaction mechanisms and the difficulties caused by the trivalent reaction medium in electrolytes, electrodes, and electrode-electrolyte interfaces. 1 (b)), aluminum anode, air-breathing cathode, and separator) can be employed with aqueous or ionic liquid electrolytes.In this manuscript, we refer to primary AAB designs in aqueous electrolytes, thus the cathode is the positive electrode, where the oxygen reduction reaction (ORR) occurs, whereas the anode is the negative electrode where Al oxidation. A newly designed aluminum-air cell is used to. The invention discloses an aluminum-air battery cathode catalyst and a preparation method thereof, wherein the preparation method comprises the steps of adding an Ag source compound, a Ni. 
Anode: + + (E 0 1.25 V) Fluid: + +Cathode: + + (E 0 0.34 V pH11) Overall + (E 0 1.59 V) Zincair batteries cannot be used in a sealed battery holder since some air must come in the oxygen in 1 liter of air is required for every ampere-hour of capacity used. Once the aluminum anode is consumed by its reaction with. Oxygen reduction reaction (ORR) activities on these air cathodes in alkaline solutions are systematic studied. The chemical equations for the zincair cell are. However, first the use of ionic liquid electrolytes leading to AlCl 4 - instead of Al 3+, the different intercalation reagents, the sluggish solid diffusion process and the fast capacity fading during cycling in aluminum batteries all need to be thoroughly explored. Al-air are primary batteries, i.e., non-rechargeable, and can also be considered to be fuel cells. At a discharge current density of 10 mA·cm2, the battery using the anolyte without water exhibited the highest specific capacity of 2328 mAh. To develop a high-capacity rechargeable aluminumair battery with resistance toward the degradation induced by long-term chargedischarge electrochemical reactions, non-oxide ceramic materials. Aluminum battery systems are considered as a system that could supplement current lithium batteries due to the low cost and high volumetric capacity of aluminum metal, and the high safety of the whole battery system. The performance of the battery was also examined. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
